Electrical patterns in the heart drive its mechanical contraction and therefore the pumping of blood. Since the cardiac muscle cells can transmit the electrical signal themselves, this pattern can go wild, leading to dangerous arrhythmias.
To understand and control complexity of excitation patterns is still challenging. In our paper, we asked ourselves: “What are the most fundamental building blocks of such a pattern, as seen on the heart’s surface?”
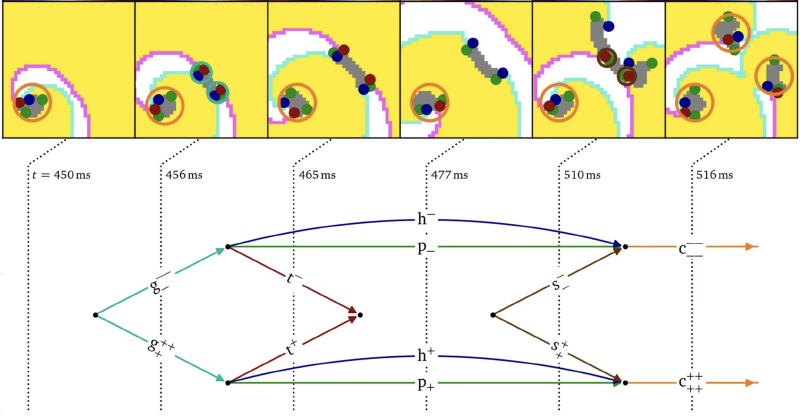
We started by considering that elementary (topological) building blocks are the regions of excited and unexcited tissue and the borders between them. Additionally, there are conduction blocks, i.e. zones where the next wave cannot yet propagate. Now, if one colors the medium in according to these 3 regions (excited, unexcited, local block), there are special points where these regions meet: heads (end points of wave fronts) and tails (end points of wave backs). These points should always be pairwise created or annihilated (with opposite chirality). Also, if one supposes that conduction blocks are thin, its end points should also appear in pairs.
The three sets of points are therefore akin to elementary particles in physics (say the 3 flavours of quarks). We propose to call these cardions, in analogy to baryons, fermions, twistons etc.
To our own surprise, we found that during ’normal’ evolution of patterns, the cardions bind together into `core particles’, which could explain why they were not observed before. During more ‘violent’ events, such as arrhythmia formation and termination, the cardions recombine.
To keep track of the topological changes during arrhythmia formation, we propose a diagrammatic way, see above. Presently, this is a schematic drawing reminiscent of the famous Feynman diagrams in physics; however, in our case there is (not yet) a deeper meaning such as the path integrals or cross-section calculations in particle physics.
Nonetheless, we show that heads, tails and pivots all have their own topological charge ($\pm$ 1/2), which constrains the possible interactions.
We are currently developing automated pipelines for cardion detection and analysis, which will enable to perform statistical analysis and systematically investigate arrhythmia initiation mechanisms.
Figure adapted from Arno et al., Scientific Reports volume 14, 28962 (2024), licensed under a Creative Commons Attribution (CC BY) license